Burkey Research Group
Organic Synthesis; Organometallic Photochemistry
The major goal of our research is the development of photochromic materials particularly those that can used in optical computers. This work by its nature is multidisciplinary and provides an opportunity for students to gain expertise in analytical, inorganic, organic, and physical chemistry and collaborate with scientists outside the Department. Activities include organic and organometallic synthesis, laser flash photolysis, high-vacuum and inert atmospheres techniques, instrument fabrication, computer programming and sample characterization by spectroscopic analysis. While each student will have an opportunity to learn and apply each of these areas specialization in one or more area is determined by the student. Students may choose among projects that provide them an opportunity to develop their problem solving skills.
- Organometallic Materials for Optical Computers
- Other Studies in the Burkey Laboratory
- Selected Publications
Organometallic Materials for Optical Computers
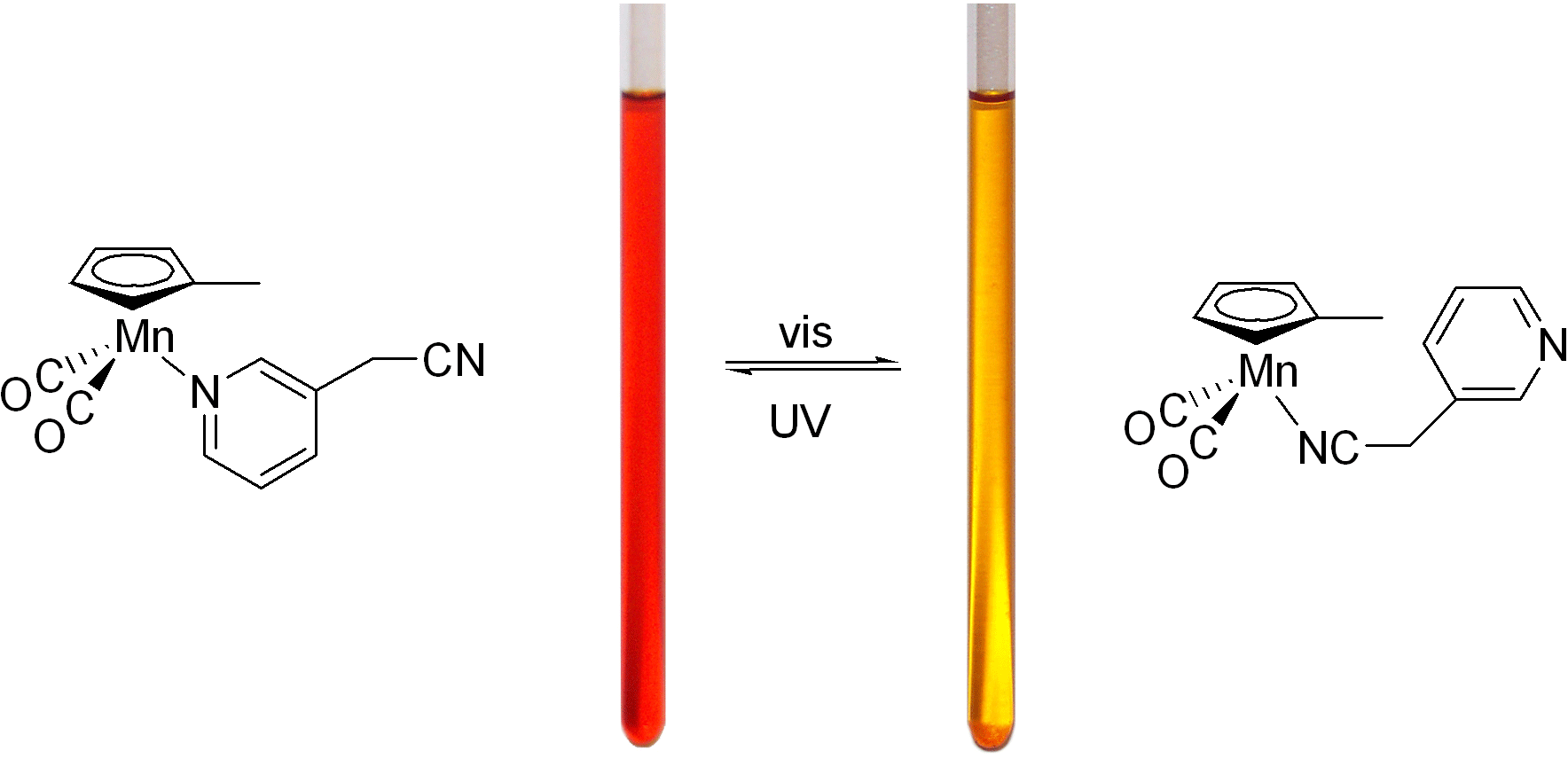
Photochromic materials are substances that change color when irradiated at one frequency and return to the original color when irradiated at another frequency. These materials can be used for data storage and if they respond rapidly could be useful for random access memory components of an optical computer. Results for our new materials suggest a potential writing speed of 107 MHz!
 Click reaction to see You Tube demo
Click reaction to see You Tube demo
The photochromic compound idealized in Scheme 1 is a long-term goal of this project.
In our model the M-L and M-L’ bonds must not be labile, except upon photolysis. Ring
closure with L’ must be exceedingly fast to compete with L recombination. A desirable
characteristic is that the product does not absorb v1, or if it does, no net chemistry occurs. The same analysis applies to the reactant
and v2. We 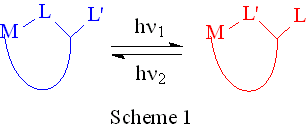 now have compounds with some of the essential features in Scheme 1. The first phase
of this work is to develop materials that have high quantum yields for ligand dissociation
and efficient ring closure. We are using organometallics for this application and
various target compounds are being synthesized and characterized for their photochromic
properties.
now have compounds with some of the essential features in Scheme 1. The first phase
of this work is to develop materials that have high quantum yields for ligand dissociation
and efficient ring closure. We are using organometallics for this application and
various target compounds are being synthesized and characterized for their photochromic
properties.
There are number of advantages to using organometallics:
1) absorption in the UV, visible and the infrared are often very strong and achievable
with low molecular weight compounds
2) metal-ligand bonds are rarely >70 kcal/mol thus high energy irradiation is unnecessary
3) hundreds of ligands may be used allowing modification of optical, thermal, physical,
chemical and kinetic properties
4) ultrafast spectroscopic methods have become available to investigate the early
steps that determine paths of excited states
For this project various target compounds are being synthesized and characterized
for their photochromic properties.
Other Studies in the Burkey Laboratory
We are undertaking several studies that support the development of photochromic organometallics.
1) In designing photochromic compounds we need to know which ligand is going to dissociates
upon photolysis; this is normally the ligand with the weakest metal-ligand bond. Bond
energies are determined in our laboratory using Laser photoacoustic calorimetry (LPAC,
see below).
2) Ring closure must be fast to compete with the recombination of the dissociated
ligand. We can determine the kinetics of ring closure and reactions with ligands by
LPAC. Some of these experiments are done in collaboration with other laboratories.
3) Different metal complexes have environmental, economic, and other practical advantages.
We survey the photochemical properties of various metal complexes.
